Write a Note on Rutherford Atomic Model Structure? OR
How Rutherford discovered that atom has a nucleus located at the center of atom?
Answer:
Rutherford performed and experiment and as a result of that experiment he discovered that nucleus located at center of atom. The experiment is known as Rutherford’s Atomic Model.
Rutherford’s Atomic Model:
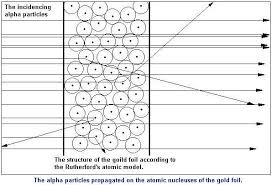
Rutherford performed “Gold Foil” experiment. He took “0.00004” thick gold foil and bombarded “alpha particles” on the foil. These alpha particles are helium nuclei (He++) and emitted from radioactive elements i.e. radium and polonium. Alpha particles can penetrate through some extent.
He observed the effect of α-particles on a photographic plate.
You see in the diagram when we through positive charge to an atom some charges are reflected from the center of the Atom which shows that positive charge is present in the center of the atom.
Result about Nucleus in Atom:
Rutherford concluded about nucleus that
- The deflection of a few particles proved that there is a “center of +ve charge” in an atom, which is called nucleus.
- The complete re-bounce of a few particles show that the nucleus is very dense and hard
- Since a few particles were deflected it shows the size of nucleus is very small
- Except electrons, all other particles lie within the nucleus are known as nucleons.
To Download this Article Click the Blue Link below:
Rutherford Atomic Model



Comments are closed.